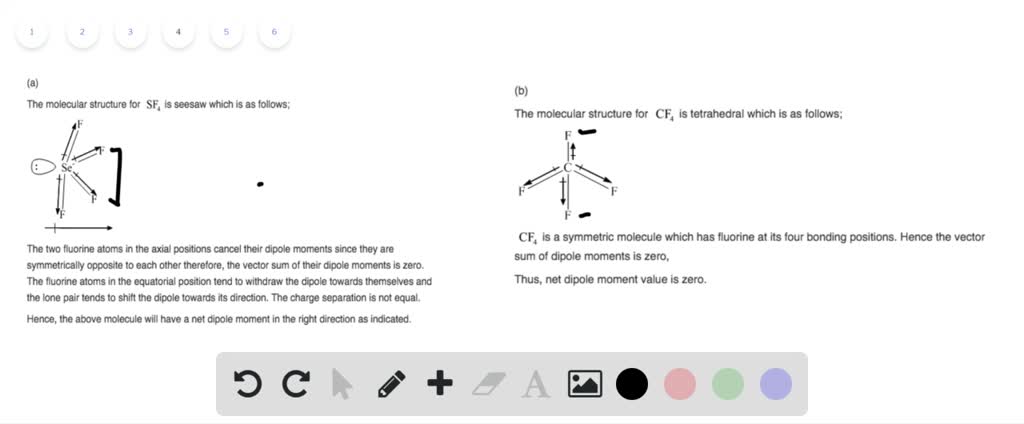
Because there is no transfer or exchange of electrons, this interaction differs from a traditional ionic or covalent bond. The two molecules become closer as a result, increasing the substance’s stability. In general, one molecule’s positive end attracts the negative end of another. The electrostatic forces between two permanent polar molecules are referred to as dipole-dipole forces or dipole-dipole interactions. 
Because molecules rotate, they tend to remain in relative orientations with low energy, such as mutual orientation with opposite partial charges adjacent to one another, the dipole–dipole interaction leads to the weak contact between molecules in gases. At low temperatures, this form of intermolecular force helps hydrogen chloride to condense into a liquid. Opposed partial charges attract one another, and if two polar molecules are oriented so that their opposite partial charges are closer together than their like charges, the two molecules will have a net attraction. The approach of the polar molecule causes a charge separation in the electron cloud of the non-polar molecule in this interaction.Ī polar molecule has an electric dipole moment due to the presence of partial charges on its atoms, as previously stated. Dipole – Dipole Forces form of intermolecular force that occurs when a polar molecule interacts with a non-polar molecule. There are various forms of intermolecular forces, and one of them is dipole-dipole forces. 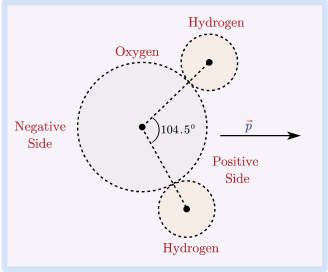
Intermolecular interactions are necessary for a molecule’s stability.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
